The increasing expenditure on health due to the pandemic, along with many circulars and regulations that support domestic pharmaceutical businesses to compete with imported products, are favorable conditions for the pharmaceutical industry to grow in the coming time.
According to a report by SSI Research, in 2020, the Pharmaceutical industry witnessed many significant changes due to the COVID-19 pandemic, such as the shortage of active pharmaceutical ingredients (API) from China and India, leading to higher drug production costs.
Social distancing and strict control of hospital visits disrupted the need for health care. As people with non-urgent health problems hesitated to do medical check-ups, there was a decrease in prescription and drug sales.
In addition, the new supply of materials in the country was interrupted due to delayed progress by new GMP factories. Due to the fact that new domestic drug manufacturers were behind schedule, and their factories could not be put into operation in 2020, imported drugs continued to dominate the market with approximately 58% of the market share.
However, it is said that although the pharmaceutical industry is sensitive to the pandemic, it is gradually recovering.
The COVID-19 epidemic presents a major challenge to the performance of the pharmaceutical industry, especially if the epidemic is widespread in the community. However, healthcare spending is generally essential and demands for it are only temporarily impacted. Therefore, there is a great possibility that the industry will recover in 2021.
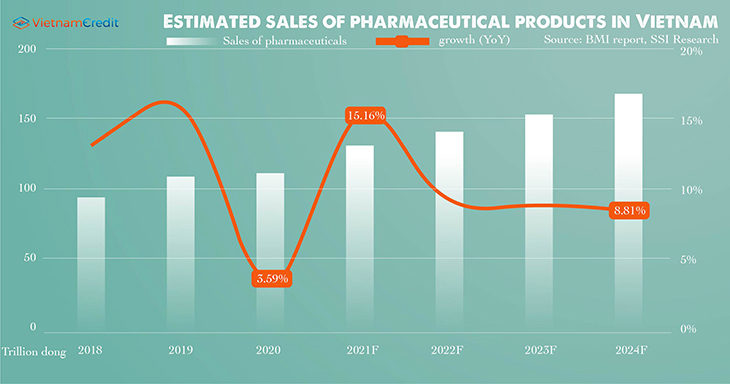
It is estimated that Vietnam's pharmaceutical revenue in 2021 will increase by 15% compared to 2020, and 2021’s CARG will be 11.8% higher than the average between 2015 and 2019’s.
Spending on health is expected return to normal in 2021 when regulations on social distancing are loosened and people's need for health care recovers.
In addition, the facts that Vietnam's population is rapidly aging and per capita income has strongly improved continue to support the long-term growth momentum of the healthcare market.
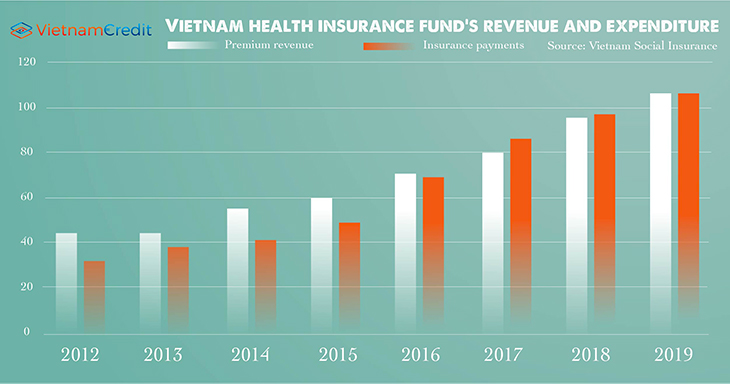
On top of that, the Government continues to amend regulations on drug bidding at public hospitals to encourage the production of generic drugs to replace expensive imported drugs and to reduce the financial burden on the health insurance fund. This is because of the fact that insurance premiums have hardly been in line with the need of insurance payment in recent years.
In 2020, the Government issued Circular 15/2020 / TT-BYT replacing Circular 09/2016 / TT-BYT, expanding the list of drugs procured by hospitals and concentrated bidding drugs (drugs directly bid by the Vietnam Social Insurance Agency).
Therefore, drug prices in hospitals are gradually becoming more transparent, helping domestic manufacturers compete with expensive imported drugs that previously dominated the hospital channel because of high commissions for medical staff.

Moreover, the Pharmaceutical Law (Chapter II, Article 7) and Circular 03/2019 / TT-BYT stipulate not to bid for imported drugs when domestically produced drugs meet the requirements of treatment, prices and supply capacity, creating a great advantage for domestic drugs.
Risks
In addition to the positive outlook for the pharmaceutical industry, there are also a number of risks such as the volatile legal environment and the fact that pharmaceutical companies heavily depend on imported API sources.
Although recent policies have supported domestic manufacturers, the legal environment for pharmaceutical companies remains unpredictable, with the amendment and enactment of 2 to 5 new circulars each year, there are many risks in doing business in this industry.
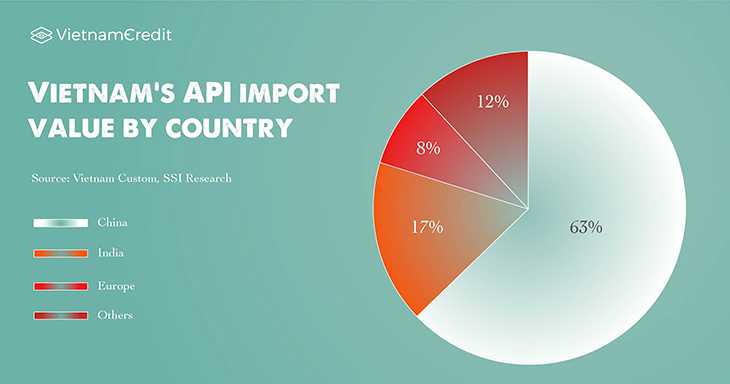
SSI Research informed that Vietnam is still heavily dependent on imported APIs from China and India, which account for 80% of the total imported APIs and 70% of the total APIs used in the industry.
This is a significant disadvantage because any changes from these countries could greatly affect the capacity of domestic drug manufacturers.
Source: SSI Research, Vietnam Custom






![[INFORGRAPHIC] 5G TECHNOLOGY: ENORMOUS POTENTIAL [INFORGRAPHIC] 5G TECHNOLOGY: ENORMOUS POTENTIAL](/Media/News/inforgraphic-5g-technology-enormous-potential.jpg)



















































